Contrast Reimbursement and Purchasing
Contrast Reimbursement and Purchasing
Reimbursement and Purchasing for Contrast Agents
Please see Full Prescribing Information, including Boxed Warnings, for Gadavist® (gadobutrol) injection, Eovist® (gadoxetate disodium) injection, and Ultravist® (iopromide) injection.
Please see Important Safety Information for Gadavist®, Eovist®, and Ultravist®.
Information related to reimbursement and purchasing for the Bayer contrast agents Gadavist® (gadobutrol), Eovist® (gadoxetate disodium), and Ultravist® (iopromide) can be found below.
If you have a question about reimbursement, please contact the Bayer Radiology Helpline at 1-800-423-7539.
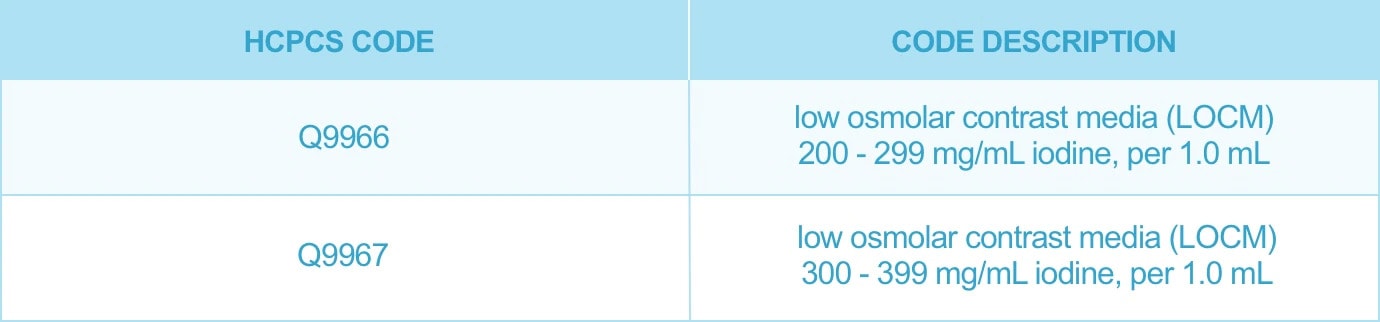
Accordion header
HCPCS code A9585 has been established by the Centers for Medicare and Medicaid Services (CMS) for Gadavist®. CMS sought to assign a code which would allow for accurate billing of Gadavist® presentations.
The code discriptor for A9585 Injection, gadobutrol, is per 0.1 mL. When billing it is important note that the unit of measure for A9585 is 0.1mL and Gadavist® dosing is calculated by mL. When billing for the dose administered, you will need to multiply the HCPCS units by 10 to be reimbursed correctly. Examples:
- 2 mL = 20 units
- 7.5 mL = 75 units
- 10 mL = 100 units
- 15 mL = 150 units
Hospital-based outpatient HCPCS code for use with Gadavist®

Freestanding HCPCS code for use with Gadavist®

Note: Information provided in this resource is for informational purposes only and does not guarantee that codes will be appropriate or that coverage and reimbursement will result. Customers should consult with their payers for all relevant coverage, coding, and reimbursement requirements. It is the sole responsibility of the provider to select proper codes and ensure the accuracy of all claims used in seeking reimbursement. Neither this resource nor the Bayer Radiology Helpline is intended as legal advice or as a substitute for a provider's independent professional judgment.
.

When billing for Eovist® for hospital outpatient services on or after January 1, 2010:
- Enter the appropriate HCPCS code, A9581, Injection gadoxetate disodium, 1 mL in record locator 44 on the CMS 1450 Form
- Enter the mL quantity of Eovist that was administered to the patient. (Unlike for Gadavist, the Eovist HPCPS code unit of measure is 1.0 mL)
Freestanding Outpatient

When billing for Eovist® for freestanding outpatient services on or after January 1, 2010:
- Enter the appropriate HCPCS code for Eovist: A9581, Injection gadoxetate disodium, 1 mL
- Enter the mL quantity of Eovist that was administered to the patient. (Unlike for Gadavist, the Eovist HPCPS code unit of measure is 1.0 mL)
To avoid billing confusion, the use of Eovist® should be noted on the patient's report dictated by the radiologist.
Note: These codes are provided for your information only. It is the responsibility of the provider to determine the most appropriate code to use in billing for services rendered. The use of these codes does not guarantee payment.
.
In today's cost-conscious healthcare environment, Bayer in Radiology offers a comprehensive line of quality imaging products at competitive prices combined with highly regarded service and support. Knowing that purchasers have unique requirements, we do our best to provide creative yet competitive contracts that go beyond just meeting product needs. Additional purchasing resources are described below.
Bayer in Radiology’s partnership with Mckesson Specialty Distribution (1-877-259-4624) combines Bayer's commitment to our customers with the drug distribution of McKesson, focused on delivering the right product to the right place at the right time. McKesson's reputation for reliable service makes them an ideal solution through which to source your Bayer imaging products.
Additional independent suppliers are listed below in alphabetical order.

INDICATIONS AND IMPORTANT SAFETY INFORMATION
IMPORTANT SAFETY INFORMATION FOR GADAVIST® (gadobutrol) AND EOVIST® (gadoxetate disodium) INJECTION
WARNING: RISK ASSOCIATED WITH INTRATHECAL USE and NEPHROGENIC SYSTEMIC FIBROSIS Risk Associated with Intrathecal Use Intrathecal administration of gadolinium-based contrast agents (GBCAs) can cause serious adverse reactions including death, coma, encephalopathy, and seizures. GADAVIST or EOVIST are not approved for intrathecal use. Nephrogenic Systemic Fibrosis GBCAs increase the risk for NSF among patients with impaired elimination of the drugs. Avoid use of GADAVIST or EOVIST in these patients unless the diagnostic information is essential and not available with non-contrasted MRI or other modalities. NSF may result in fatal or debilitating fibrosis affecting the skin, muscle and internal organs. The risk of NSF appears highest among patients with:
Screen patients for acute kidney injury and other conditions that may reduce renal function. For patients at risk for chronically reduced renal function (for example, age >60 years, hypertension or diabetes), estimate the glomerular filtration rate (GFR) through laboratory testing. For patients at highest risk for NSF, do not exceed the recommended GADAVIST or EOVIST dose and allow a sufficient period of time for elimination of the drug from the body prior to any re-administration. |
ADDITIONAL IMPORTANT SAFETY INFORMATION FOR GADAVIST®
Contraindication and Important Information about Hypersensitivity Reactions: Gadavist® is contraindicated in patients with history of severe hypersensitivity reactions to Gadavist®. Anaphylactic and other hypersensitivity reactions with cardiovascular, respiratory, or cutaneous manifestations, ranging from mild to severe, have occurred following Gadavist® administration. There have been reports of life-threatening and fatal outcomes from these adverse reactions. Before Gadavist® administration, assess all patients for any history of a reaction to contrast media, bronchial asthma and/or allergic disorders. These patients may have an increased risk for a hypersensitivity reaction to Gadavist®.
Acute Respiratory Distress Syndrome (ARDS): ARDS has been reported with Gadavist® and may be characterized by severe hypoxemia requiring oxygen support and mechanical ventilation. Onset can occur within < 30 minutes to 24 hours after administration. For patients demonstrating respiratory distress after administration, assess oxygen requirement and monitor for worsening respiratory function.
Gadolinium Retention: Gadolinium is retained for months or years in several organs. Linear GBCAs cause more retention than macrocyclic GBCAs. At equivalent doses, retention varies among the linear agents. Retention is lowest and similar among the macrocyclic GBCAs. Consequences of gadolinium retention in the brain have not been established, but they have been established in the skin and other organs in patients with impaired renal function. While clinical consequences of gadolinium retention have not been established in patients with normal renal function, certain patients might be at higher risk. These include patients requiring multiple lifetime doses, pregnant and pediatric patients, and patients with inflammatory conditions. Consider the retention characteristics of the agent and minimize repetitive GBCA studies, when possible.
Acute Kidney Injury: In patients with chronic renal impairment, acute kidney injury sometimes requiring dialysis has been observed with the use of GBCAs. Do not exceed the recommended dose; the risk of acute kidney injury may increase with higher than recommended doses.
Extravasation and Injection Site Reactions: Extravasation into tissues during Gadavist® administration may result in moderate irritation. Ensure catheter and venous patency before injection.
Overestimation of Extent of Malignant Disease in MRI of the Breast:Gadavist® MRI of the breast overestimated the histologically confirmed extent of malignancy in the diseased breast in up to 50% of the patients.
Low Sensitivity for Significant Arterial Stenosis: The performance of Gadavist® MRA for detecting arterial segments with significant stenosis (>50% renal, >70% supra-aortic) has not been shown to exceed 55%. Therefore, a negative MRA study alone should not be used to rule out significant stenosis.
Adverse Reactions: The most frequent (≥0.5%) adverse reactions associated with Gadavist® in clinical studies were headache (1.7%), nausea (1.2%) and dizziness (0.5%).
Please see Full Prescribing Information for Gadavist®
GADAVIST® INDICATIONS
Gadavist® is indicated for:
- Magnetic resonance imaging (MRI) of the central nervous system (CNS) to detect and visualize areas with disrupted blood brain barrier and/or abnormal vascularity in adult and pediatric patients including term neonates
- MRI of the breast to assess the presence and extent of malignant breast disease in adult patients
- Magnetic resonance angiography (MRA) to evaluate known or suspected supra-aortic or renal artery disease in adult and pediatric patients including term neonates
- Cardiac MRI (CMRI) to assess myocardial perfusion (stress, rest) and late gadolinium enhancement in adult patients with known or suspected coronary artery disease (CAD)
ADDITIONAL IMPORTANT SAFETY INFORMATION FOR EOVIST®
Contraindication and Important Information about Hypersensitivity Reactions: Eovist® is contraindicated in patients with history of severe hypersensitivity reactions to Eovist®. Anaphylactic and other hypersensitivity reactions with cardiovascular, respiratory and cutaneous manifestations, ranging from mild to severe, including shock have occurred following Eovist® administration. Before Eovist® administration, assess all patients for any history of a reaction to contrast media, bronchial asthma and allergic disorders. These patients may have an increased risk for a hypersensitivity reaction to Eovist®.
Gadolinium Retention: Gadolinium is retained for months or years in several organs. Linear GBCAs cause more retention than macrocyclic GBCAs. At equivalent doses, retention varies among the linear agents. Retention is lowest and similar among the macrocyclic GBCAs. Consequences of gadolinium retention in the brain have not been established, but they have been established in the skin and other organs in patients with impaired renal function. While clinical consequences of gadolinium retention have not been established in patients with normal renal function, certain patients might be at higher risk. These include patients requiring multiple lifetime doses, pregnant and pediatric patients, and patients with inflammatory conditions. Consider the retention characteristics of the agent and minimize repetitive GBCA studies, when possible.
Acute Kidney Injury: In patients with chronic renal impairment, acute kidney injury sometimes requiring dialysis has been observed with the use of GBCAs.Do not exceed the recommended dose; the risk of acute kidney injury may increase with higher than recommended doses.
Extravasation and Injection Site Reactions: Extravasation into tissues during Eovist® administration may result in local tissue reactions such as myocyte necrosis and inflammation. Ensure catheter and venous patency before the injection.
Interference with Laboratory Tests: Serum iron determination using complexometric methods may result in falsely high or low values for up to 24 hours after the examination with Eovist®.
Interference with Visualization of Liver Lesions: End stage renal failure or hepatic failure may impair Eovist® imaging performance. In patients with elevated serum ferritin or serum bilirubin >3 mg/dL, reduced hepatic contrast was observed.
Adverse Reactions: The most frequent (≥0.5%) adverse reactions associated with Eovist® are nausea (1.1%), headache (1.1%), feeling hot (0.8%), dizziness (0.6%), and back pain (0.6%).
Please see Full Prescribing Information for Eovist®.
EOVIST® INDICATION
Eovist® is indicated for use in magnetic resonance imaging (MRI) of the liver to detect and characterize lesions in adult and pediatric patients, including term neonates, with known or suspected focal liver disease.
ULTRAVIST® (iopromide) injection
IMPORTANT SAFETY INFORMATION
WARNING: RISK ASSOCIATED WITH INTRATHECAL USE
Intrathecal administration, even if inadvertent, may cause death, convulsions, cerebral hemorrhage, coma, paralysis, arachnoiditis, acute renal failure, cardiac arrest, seizures, rhabdomyolysis, hyperthermia, and brain edema. Ultravist® is not approved for intrathecal use.
Risks Associated with Intrathecal Use: Intrathecal administration, even if inadvertent, can cause death, convulsions, cerebral hemorrhage, coma, paralysis, arachnoiditis, acute renal failure, cardiac arrest, seizures, rhabdomyolysis, hyperthermia, and brain edema. Ultravist® is for intra-arterial or intravenous use only. Ultravist® is not approved for intrathecal use.
Hypersensitivity Reactions: Ultravist® can cause life-threatening or fatal hypersensitivity reactions including anaphylaxis. Manifestations include respiratory arrest, laryngospasm, bronchospasm, angioedema, and shock. Most severe reactions develop shortly after the start of injection (e.g., within 1 to 3 minutes), but delayed reactions can also occur. There is increased risk of hypersensitivity reactions in patients with a history of previous reaction to a contrast agent and known allergic disorders, or other hypersensitivities. Premedication with antihistamines or corticosteroids does not prevent serious life-threatening reactions but may reduce both their incidence and severity. Obtain a history of allergy, hypersensitivity, or hypersensitivity reactions to iodinated contrast agents and have emergency resuscitation equipment and trained personnel available prior to Ultravist® administration. Monitor all patients for hypersensitivity reactions.
Acute Kidney Injury: Acute kidney injury, including renal failure, may occur after administration. Risk factors include: pre-existing renal insufficiency, dehydration, diabetes mellitus, congestive heart failure, advanced vascular disease, elderly age, concomitant use of nephrotoxic or diuretic medications, multiple myeloma or other paraproteinemia, and repetitive and/or large doses of ®. Use the lowest necessary dose of® in patients with renal impairment. Hydrate patients prior to and following ® administration. Do not use laxatives, diuretics, or preparatory dehydration prior to® administration.
Cardiovascular Adverse Reactions: Acute or delayed hemodynamic disturbances may occur in patients with congestive heart failure (CHF), severe renal dysfunction, combined renal and hepatic disease, or combined renal and cardiac disease, particularly with repetitive and/or large doses. Fatal cardiovascular reactions have occurred mostly within 10 minutes of Ultravist® injection. Hypotensive collapse and shock have occurred. Cardiac decompensation, serious arrhythmias, and myocardial ischemia or infarction can occur during coronary arteriography and ventriculography. Use the lowest necessary dose of Ultravist® in patients with CHF. Always have emergency resuscitation equipment and trained personnel available. Monitor all patients for severe cardiovascular reactions.
Thromboembolic Events: Serious, in some cases fatal, thromboembolic events causing myocardial infarction and stroke can occur during angiography procedures. During these procedures, increased thrombosis and activation of the complement system can occur. Risk of thromboembolic events can be influenced by: length of procedure, catheter and syringe material, underlying disease state, and concomitant medications. To decrease thromboembolic events, use meticulous angiographic techniques and minimize the length of the procedure. Avoid blood remaining in contact with syringes containing iodinated contrast agents, which increases the risk of clotting. Avoid angiography in patients with homocystinuria because of the risk of inducing thrombosis and embolism.
Extravasation and Injection Site Reactions: Extravasation can occur, particularly in patients with severe arterial or venous disease. In addition, injection site reactions such as pain and swelling at the injection site can also occur. Ensure intravascular placement of catheters prior to injection. Monitor patients for extravasation and advise patients to seek medical care for progression of symptoms.
Thyroid Storm in Patients with Hyperthyroidism: Thyroid storm has occurred after the intravascular use of iodinated contrast agents in patients with hyperthyroidism or with an autonomously functioning thyroid nodule. Evaluate the risk in such patients before use of Ultravist®.
Thyroid Dysfunction in Pediatric Patients 0 to 3 Years of Age: Thyroid dysfunction characterized by hypothyroidism or transient thyroid suppression has been reported after both single exposure and multiple exposures to iodinated contrast media (ICM) in pediatric patients 0 to 3 years of age. Younger age, very low birth weight, prematurity, underlying medical conditions affecting thyroid function, admission to neonatal or pediatric intensive care units, and congenital cardiac conditions are associated with an increased risk of hypothyroidism after ICM exposure. After exposure to ICM, individualize thyroid function monitoring based on underlying risk factors, especially in term and preterm neonates. The safety and effectiveness of Ultravist® in pediatric patients younger than 2 years of age have not been established, and Ultravist® is not approved for use in pediatric patients younger than 2 years of age.
Hypertensive Crisis in Patients with Pheochromocytoma: Hypertensive crisis in patients with pheochromocytoma has occurred with iodinated contrast agents. Closely monitor patients when administering Ultravist® if pheochromocytoma or catecholamine-secreting paragangliomas are suspected. Inject the minimum amount of Ultravist® necessary and have measures for treatment of a hypertensive crisis readily available.
Sickle Cell Crisis in Patients with Sickle Cell Disease: Iodinated contrast agents may promote sickling in individuals who are homozygous for sickle cell disease. Hydrate patients prior to and following administration and use only if the necessary imaging information cannot be obtained with alternative imaging modalities.
Severe Cutaneous Adverse Reactions: Severe cutaneous adverse reactions (SCAR) may develop from 1 hour to several weeks after intravascular contrast agent administration. These reactions include Stevens-Johnson syndrome and toxic epidermal necrolysis (SJS/TEN), acute generalized exanthematous pustulosis (AGEP), and drug reaction with eosinophilia and systemic symptoms (DRESS). Reaction severity may increase and time to onset may decrease with repeat administration of contrast agent; prophylactic medications may not prevent or mitigate severe cutaneous adverse reactions. Avoid administering Ultravist® to patients with a history of a severe cutaneous adverse reaction to Ultravist®.
Interference with Laboratory Tests: Ultravist® can interfere with protein-bound iodine test.
Common Adverse Reactions: Common adverse reactions (>1%) are headache, nausea, injection site and infusion site reactions, vasodilatation, vomiting, back pain, urinary urgency, chest pain, pain, dysgeusia, and abnormal vision.
ULTRAVIST® INDICATIONS
Intravenous Procedures*: Ultravist® is indicated for:
- Contrast Computed Tomography (CT) of the head and body (intrathoracic, intra-abdominal, and retroperitoneal regions) for the evaluation of neoplastic and non-neoplastic lesions in adults and pediatric patients aged 2 years and older
- Contrast mammography to visualize known or suspected lesions of the breast in adults, as an adjunct following mammography and/or ultrasound
- Excretory urography in adults and pediatric patients aged 2 years and older.
Intra‐arterial Procedures*: Ultravist® is indicated for:
- Cerebral arteriography and peripheral arteriography in adults
- Coronary arteriography and left ventriculography, visceral angiography, and aortography in adults
- Radiographic evaluation of cardiac chambers and related arteries in pediatric patients aged 2 years and older.
*Specific concentrations and presentations of Ultravist® are recommended for each type of imaging procedure (see Dosage and Administration 2.2, 2.3, 2.4 in the Full Prescribing Information).